Electron Microscopy in Life Science
Electron microscopy is a common approach for examining life at the nanoscale. In life sciences, it helps biologists to examine cells, cellular processes, and organelle architecture at nanometer resolution. For example, in cell biology it is used to provide answers by studying the interplay between macromolecules, organelles and cells. Electron microscopy is also useful, and potentially the only possible approach, in the field of connectomics for visualising and reconstructing neurons and their interactions.
Limitations of Electron Microscopy – Acquisition Speed
Nevertheless, as powerful as electron microscopy can be, it is still limited in many ways. For example, data is often collected out of context due to typically small fields of view that can be acquired in a given timeframe. The current status of electron microscopy is that it is indeed a very slow approach. In the field of neurobiology, for instance, collecting images to reconstruct relevant volumes for the field, e.g. 0.5 x 0.5 x 0.5 mm of material with a resolution of 4 x 4 x 40 nm, would require years to complete. In other fields, typically electron microscopy is used as a qualitative tool, because acquiring enough information to make statistically relevant conclusions is onerous from a time perspective.
FAST-EM – Large Volume Electron Microscopy for Life Science
To overcome these bottlenecks, Delmic developed FAST-EM, an ultra-fast scanning transmission electron microscope specifically designed and optimised for automating the workflow of large volume electron microscopy (EM) for life science. The system was developed together with Thermo Fisher Scientific, Technolution, and TU Delft with the main mission of simplifying and accelerating EM workflows to enable large volume acquisitions in complete autonomy.
How Large Volume Electron Microscopy is Achieved
How is the optimised EM workflow achieved? The system uses 64 individual beams that scan in parallel, which makes the image acquisition much faster. The beams are created from a Schottky field emission source with a custom aperture lens array with 64 holes. The beams are scanned in parallel over the sample in a square grid, so that each beamlet images a small part of the sample. The result is processed into a single seamless image. The method used by FAST-EM to acquire high-resolution images is transmission detection. By monitoring the quality of the 64 beamlets with a diagnostic camera and automatically adjusting focus, stigmation, and alignment when required during the acquisition, long acquisition runs can be performed in complete autonomy, unattended.

The system can acquire large areas autonomously and present entire regions of interest, pre-stitched, on tools such as CATMAID. The typical volume of 0.5 x 0.5 x 0.5 mm can be acquired by FAST-EM in just 22 days of continuous operation, as opposed to years required by a conventional system. As another example, the image provided here can be acquired in just below 10 seconds, as opposed to several minutes required by a standard SEM.
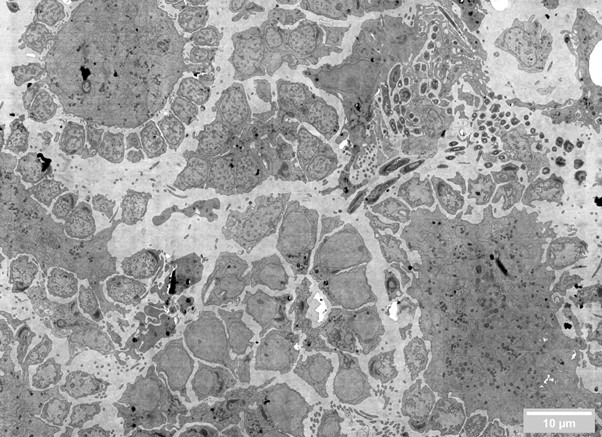
The substrates of FAST-EM are compatible with most sample preparation and collection techniques, from more manual approaches to more automated ones such as the Leica Artos 3D system [1], or the Collectome MagC system [2].
Summary
FAST-EM is the next generation electron microscope that can bring you from sample preparation to insight with unprecedented speed.
References
[1] https://www.leica-microsystems.com/products/sample-preparation-for-electron-microscopy/p/artos-3d/
[2] Templier, T., 2019. MagC, magnetic collection of ultrathin sections for volumetric correlative light and electron microscopy. University of Zurich and ETH Zurich, Switzerland. eLife

